
You will need to refer to a periodic table for proton values. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Why? Because they share properties that are considered to be a cross between non-metals and metals.\nonumber \] 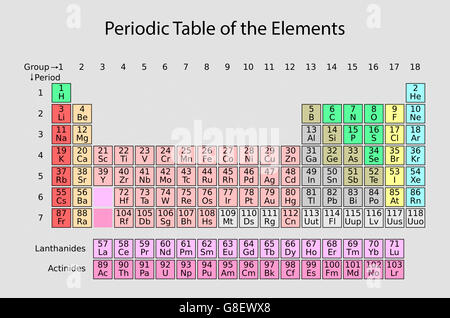
Some of the elements of the Periodic table are classified as semimetals or metalloids. Remember that the only liquid metal element is Mercury (Hg). And, instead of easily losing electrons as metals do, non-metals gain them through chemical reactions.Īpart from this, some non-metals are liquid. And they are not ductile (you cannot make them into thin wires) or malleable (they can not be made into thin sheets. So, non-metals are brittle instead of solid. Non-metals are characterized by having the exact opposite properties of metals. Li Name of Element : Lithium Atomic Weight : 6. He Name of Element : Helium Atomic Weight : 4.0 Atomic Number : 2 Group : Nobel Gases Electron Configuration: 1s2 Go to the Top of the page. The only exception to this is atomic number 1, Hydrogen (H), which has a different location on the table. H Name of Element : Hydrogen Atomic Weight : 1.0 Atomic Number : 1 Group : Non-Metals Electron Configuration: 1s1 Go to the Top of the page. Non-metals can be easily located on the Periodic Table because they are to the right of the line that looks like a stepping ladder. – Sam Kean Non-Metals In The Periodic Table But the symbol for mercury, Hg, consists of two letters that don’t even appear in its name. It is there – between gold, which is also dense and soft, and thallium, which is also poisonous. When first presented with the jumble of the periodic table, I scanned for mercury and couldn’t find it. Here is the complete list of metal elements, with the whole names, symbols, and their atomic numbers in the Periodic Table: Element I still have a vivid memory of my excitemet when I first saw a chart of the periodic table of elements. And they all can also be made into very thin sheets, which is what makes them malleable.Īside from those features, metals are also characterized by their ability to easily lose electrons. All metals can be made into thin wires, this feature is technically known as ductility. All metals conduct heat and electricity, and they all have a shiny appearance. 4 The JWP has earlier reported 5-7 on the discovery of elements with atomic numbers 112, 114, and 116. 
2016 preprint 2 recently published in Chemistry International. Metals are solid, with the sole exception of Mercury (Hg), which is liquid. Names and symbols of the elements 113, 115, 117 and 118 Pure Appl. They all share all or most of the following features: That way it frees up some part of your brain to do something else.” - Bill Nyeīut, what are metals? The elements that are classified as metals all share a number of characteristics. Atomic Number Chemical Symbol Element Name Atomic Weight (u) 1: H: Hydrogen: 1.008: 2: He: Helium: 4.002602: 3: Li: Lithium: 6.94: 4: Be: Beryllium: 9. “If you memorize the periodic table it will speed you up if you’re a chemist, but by and large, the reason you have a periodic table is so that you can store that information outside of your body. Everything else to the left of those elements is classified as metal. There are only two exceptions, i.e., two elements in that sequence (between number 5 and number 84) that are not metals: atomic number 32, Germanium (Ge) and atomic number 52, Antinomy (Sb). If you look at the Periodic table you will find that the metal elements are located between atomic number 5, Boron (B) all the way to atomic number 84 Polonium (Po). 
So, because most elements of the Table are metals it makes sense to begin by looking at them. Let’s dive into the location of both metals and nonmetals in the periodic table! Metals In The Periodic Table If you do that, you will soon notice that most elements of the Periodic Table are metals, however, a few nonmetals are scattered about. One of the best ways to classify the elements is into metals and non-metals. Using it, you should be able to classify all the elements in different ways. The Periodic Table contains a lot of useful information on the elements. Metals, nonmetals, and metalloids make up the periodic table, with metals constituting the large majority of all metals. The periodic table of metals and nonmetals can be broken down to give you a sense of each element’s characteristics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
